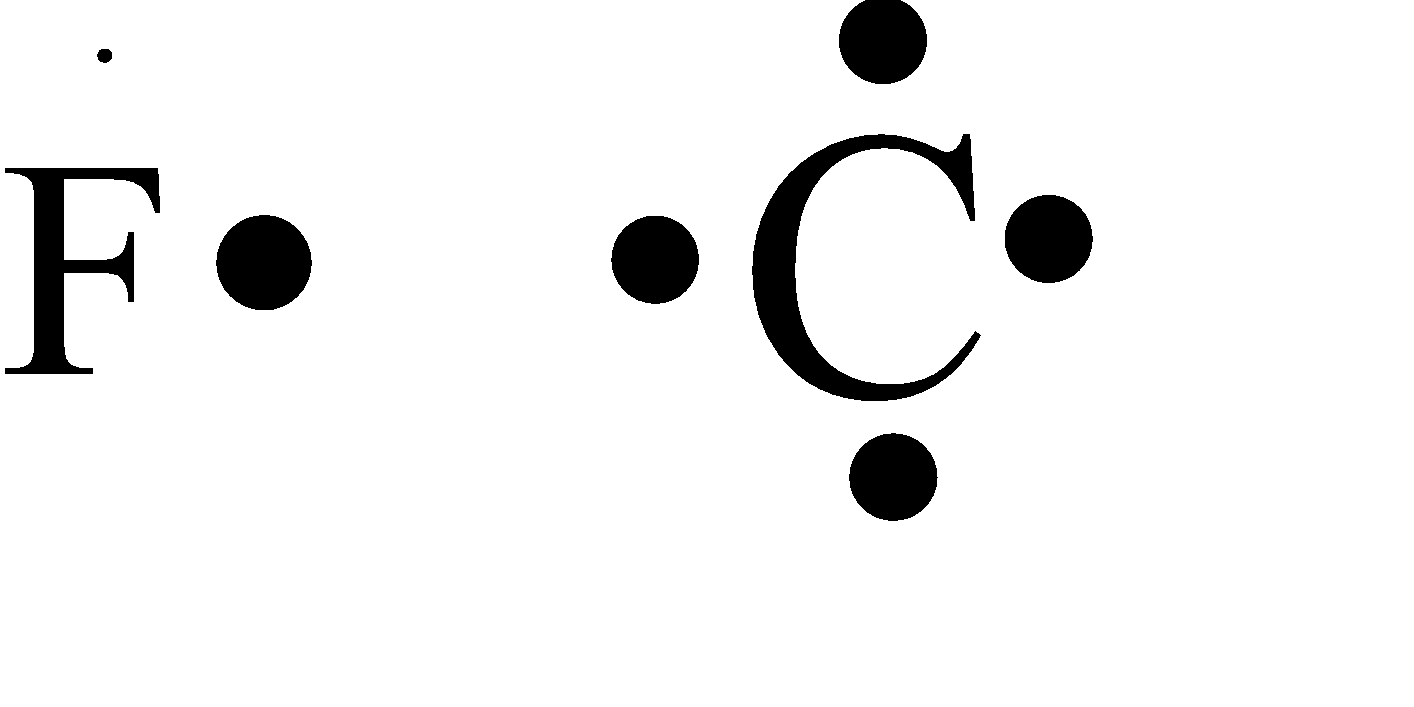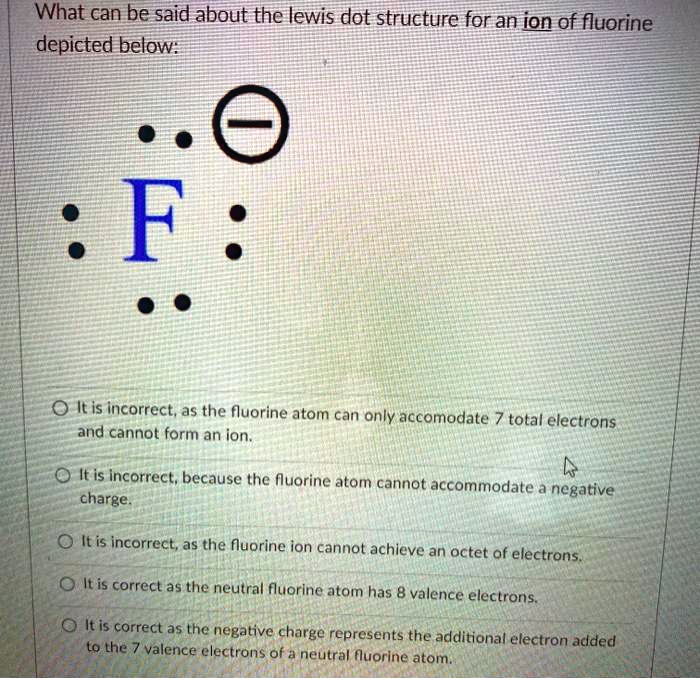
SOLVED: What can be said about the Lewis dot structure for an ion of fluorine depicted below: F It is incorrect, as the fluorine atom can only accommodate 7 total electrons and

periodic table - How to know the number of valence electrons of f-block elements? - Chemistry Stack Exchange

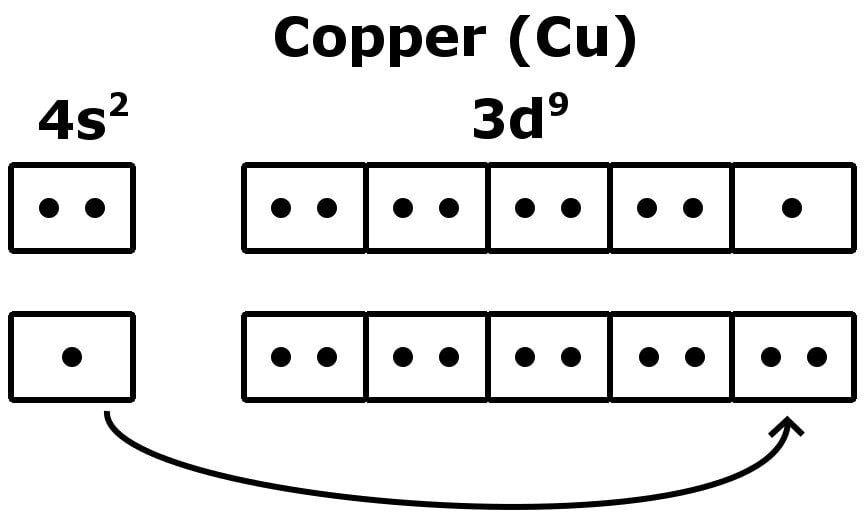
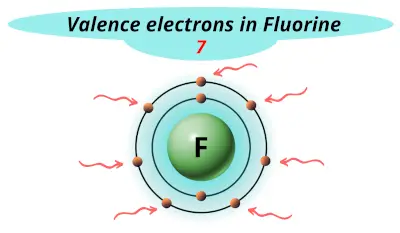
Fluorine (F) Orbital diagram, Electron configuration, and Valence electrons | Electron configuration, Electrons, Configuration