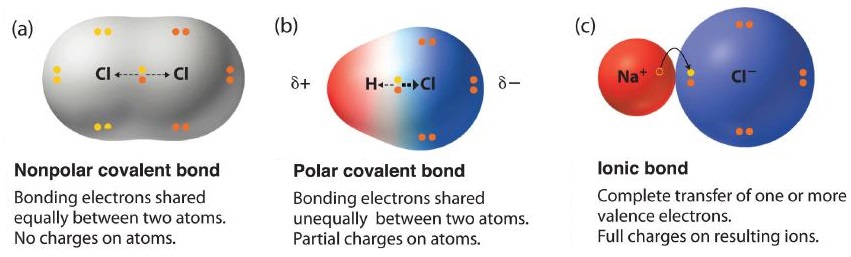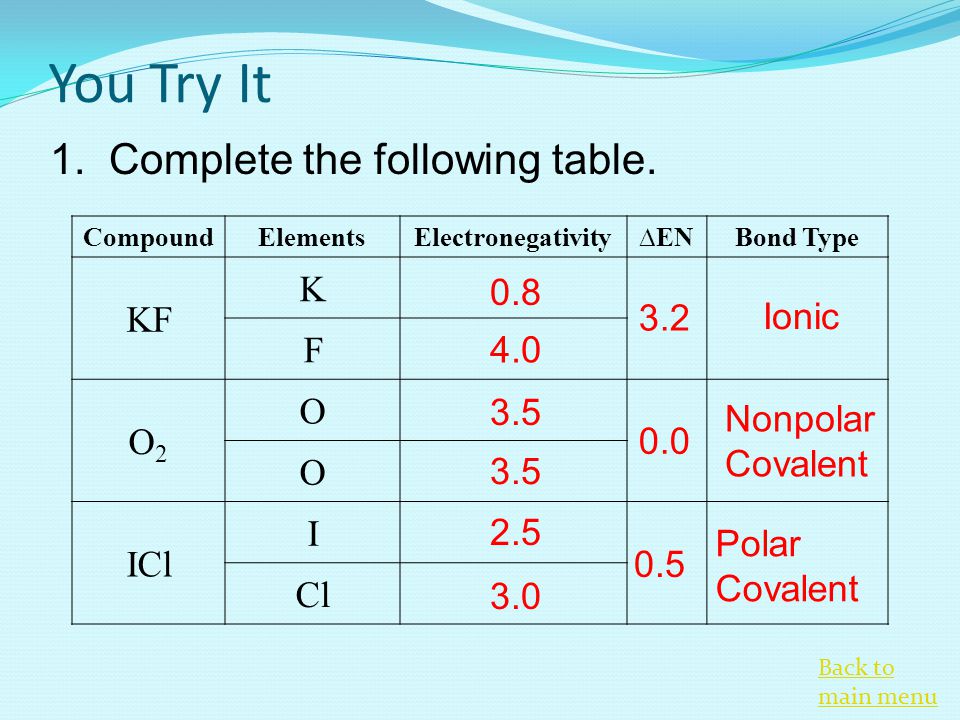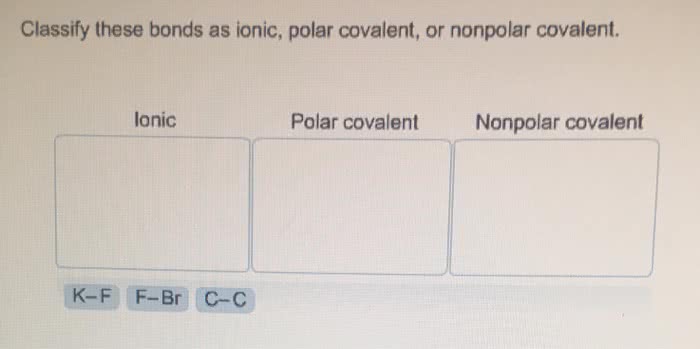
OneClass: Classify these bonds as ionic, polar covalent, or nonpolar covalent. lonic Polar covalent N...

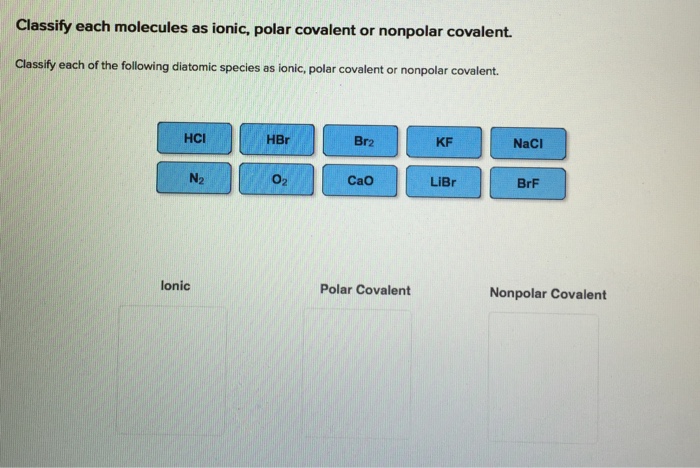
Classify each of the following diatomic species as ionic, polar covalent or nonpolar covalent. | Homework.Study.com

SOLVED: Differentiate between what happens when the following are added to water a. polar solute versus nonpolar solute b. KF versus C6H12O6 c. RbCl versus AgCl d. HNO3 versus CO

For each solute, identify the better solvent: water or carbon tetrachloride. Water, H2O Carbon tetrachloride - Home Work Help - Learn CBSE Forum

HYDRANAL™ - LipoSolver CM, Reagent for volumetric one-component KF titration in non-polar substances, fats

The following molecules contain polar covalent bonds. Which of them are polar molecules? 1. H2S 2. CCl4 3. CO2 4. SO2 5. CHCl3 | Homework.Study.com
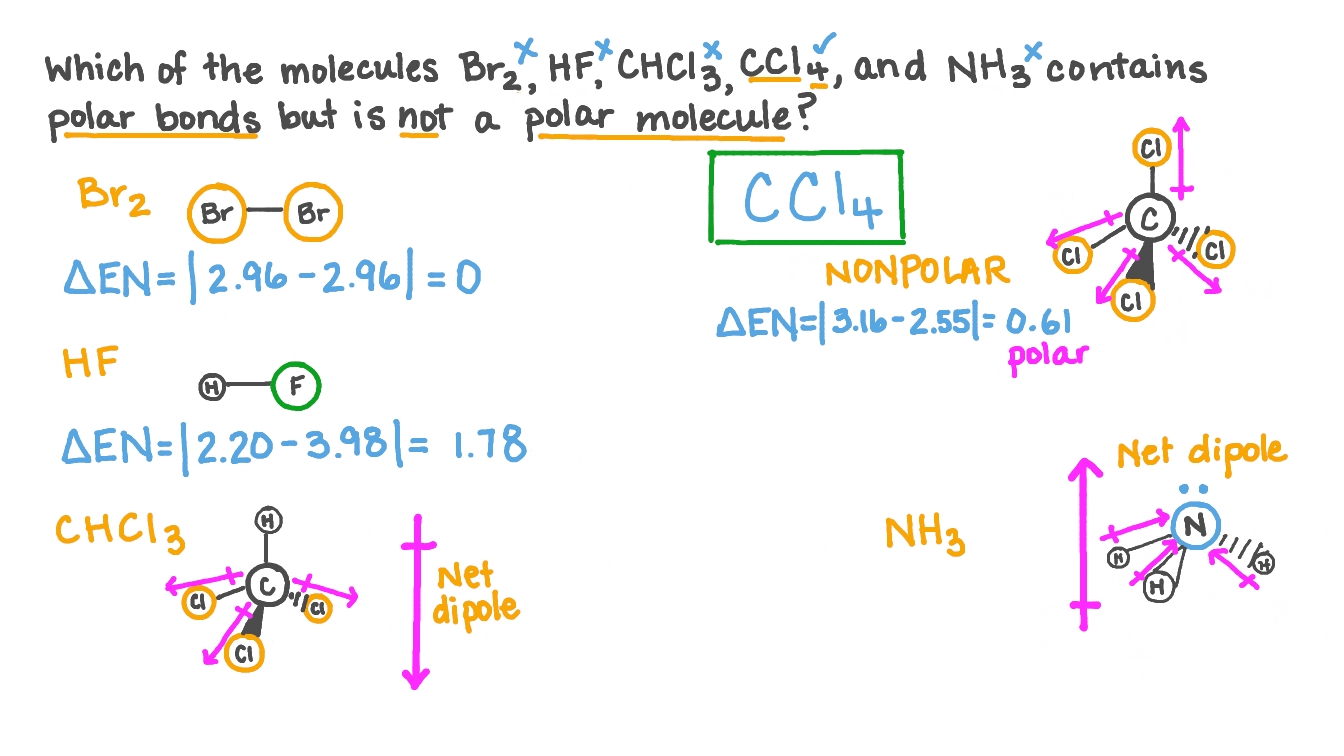
Question Video: Determining the Molecule That Contains Polar Bonds but Is Not a Polar Molecule | Nagwa
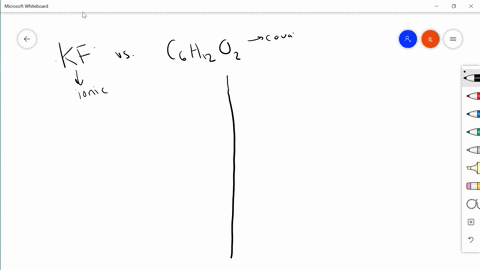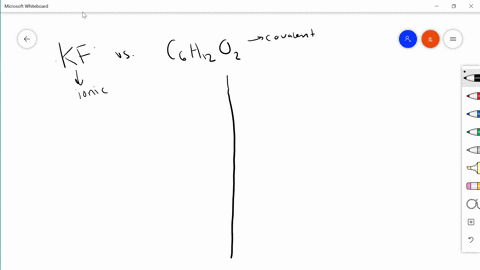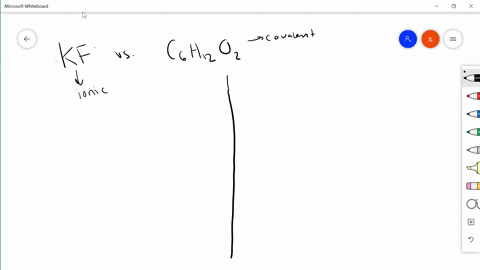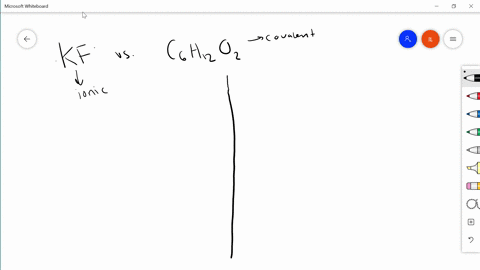
SOLVED:Differentiate between what happens when the following are added to water. a. polar solute versus nonpolar solute b. KF versus C6 H12 O6 c. RbCl versus AgCl d. HNOversus CO
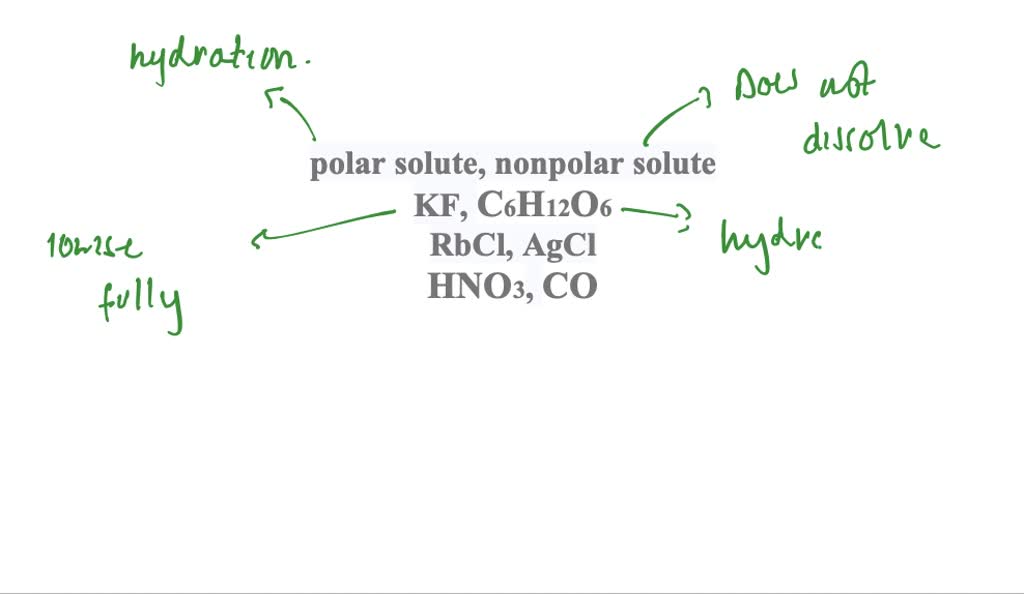
SOLVED:Differentiate between what happens when the following are added to water a. polar solute versus nonpolar solute b. KF versus C6 H12 O6 c. RbCl versus AgCl d. HNO3 versus CO